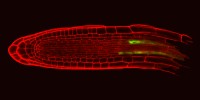
Masle Group - Environmental sensing, systemic signalling and development: plant resilience to drought and changing climate
Plants are sessile and exposed to dynamic, highly variable environmental and often stressful conditions, below and above-ground. In order to establish, survive, effectively compete with neighbours, grow and produce sustained yield and edible food, they have evolved an astonishing plasticity in the way they organise, structure, size and deploy their organs in space and time.
While long documented and the object of fascination and intense interest from naturalists and evolutionary biologists, how this developmental plasticity is controlled and integrated from the molecular to whole plant levels and phenotypically expressed remains little understood. A picture has emerged over recent years where plants are equipped of highly sensitive and coordinated mechanisms integrating developmental and environmental signals and mounting appropriate functional and developmental responses. Developmental plasticity and functional plasticity are intricately linked. Understanding how this is achieved is recognised as one of the major challenges in plant biology research, made even more pressing in the context of climate change. Contributing this ambitious goal underpins our research.
Our central interest is on the interplay between genetic and environmental determinants of plant development and adaptation to stress. We are studying how plants sense their environment and integrate endogenous controls and environmental cues to modulate their shape, size and architecture, and make optimum use of available resources for growth and survival.
Although our focus is on plants, we exploit the facinating overlaps that exist at the molecular level in developmental and stress response networks across multi-cellular organisms, and several of our projects cut across research in humans for example. We work on a range of plant species, from model species to crop/horticultural species or their wild relative, depending on the question posed, and combine physiology, developmental biology, biochemistry, genomic approaches.
Roots as sensors and signalling agents to aerial meristems, expanding leaves and stomata.
Master genes for inter-organ coordination of plant development and plasticity under adverse conditions.
Leaf patterning, leaf surface characteristics and intercellular communication for an optimal trade-off between carbon gain and water loss.
Environmental sensing in seeds: germinate or remain quiescent? Molecular controls; adaptive significance.
Tapping into the gene pool of tomato wild-relatives to improve drought resistance.
A wide range of opportunities are available for research projects within the lab, from short term undergraduate research projects to Honours, Masters and PhD projects.
If you are interested, contact josette.masle@anu.edu.au Our projects can be tailored to a wide range of interests.
| Project | Status |
|---|---|
| Abiotic stress signalling, developmental programming and plant adaptation | Current |
Selected publications
-
Rashid FAA, Crisp PA, Zhang Y, Berkowitz O, Pogson BJ, Day DA, Masle J, Dewar RC, Whelan J, Atkin OK, Scafaro AP. 2020 Molecular and physiological responses during thermal acclimation of leaf photosynthesis and respiration in rice. 2020. Plant Cell & Environment 43:594-610. https://doi.org/10.1111/pce.13706
Hossain SM, Masle J, Easton A , Hunter MN, Godwin ID, Farquhar GD, Lambrides CJ. 2020. Genetic variation for leaf carbon isotope discrimination and its association with transpiration efficiency in canola (Brassica napus). Functional Plant Biology 4:355-367 https://doi.org/10.1071/FP19256.
Rashid FAA, Scafaro AP, Asao S, Fenske R, Dewar RC, Masle J, Taylor NL, Atkin OK. 2020. Diel‐ and temperature‐driven variation of leaf dark respiration rates and metabolite levels in rice. New Phytologist https://doi.org/10.1111/nph.16661
- Nanda AK, El Habti A, Hocart C, Masle J. 2019. Timing of seed germination under changing salinity: a key role of the ERECTA receptor-kinases. Journal of Experimental Botany, doi: 10.1093/jxb/erz385
- Branco R, Masle J. 2019. Systemic signalling through TCTP1 controls lateral root formation in Arabidopsis. Journal of Experimental Botany, 70:3927–3940, https://doi.org/10.1093/jxb/erz204
- Chen W, Taylor MC, Barrow RA, Croyal M, Masle J. 2019. Loss of Phosphoethanolamine N-Methyltransferases abolishes phosphatidylcholine synthesis and is lethal. Plant Physiology, https://doi.org/10.1104/pp.18.00694.
- Chen W, Salari H, Taylor MC, Jost R, Berkowitz O, Barrow R, Qiu D, Branco R, Masle J. 2018. NMT1 and NMT3-N methyltransferase activity is critical to lipid homeostasis, morphogenesis and reproduction. Plant Physiology https://doi.org/10.1104/pp.18.00457.
- Cazzonelli CI, Vanstraelen M, Yin K, Carron-Arthur A, Nisar N, Tarle G, Cuttriss AJ, Searle IR, Simon S, Benkova E, Mathesius U, Masle J, Friml J, Pogson BJ. 2013. Role of the Arabidopsis PIN6 auxin transporter in auxin homeostasis and auxin-mediated development. PlosOne https://doi.org/10.1371/journal.pone.0070069
- Yun-Kuan Liang, Xiaodong Xie, Shona E. Lindsay, Yi Bing Wang, Josette Masle, Lisa Williamson, Ottoline Leyser and Alistair M. Hetherington. 2010. Cell wall composition contributes to the control of transpiration efficiency in Arabidopsis thaliana. The Plant Journal https://doi.org/10.1111/j.1365-313X.2010.04362.x
- Jost R, Berkowitz O, Shaw JE, Masle J. 2009. Biochemical characterisation of two wheat phosphoethanolamine N-methyltransferase isoforms with different sensitivities to inhibition by phosphatidic acid. Journal of Biological Chemistry, 284, 46:31962-31971.
- Berkowitz O, Jost R, Pollman S and Masle J. 2008. Characterisation of TCTP, the translationally controlled Tumor Protein, from Arabidopsis thaliana. Plant Cell, 20:3430-3447.
- Hoque MS, Masle J, Udvardi MK, Ryan PR, Upadhyaya NM. 2006. Over-expression of the rice OsAMT1-1 gene increases ammonium uptake and content, but impairs growth and development of plants under high ammonium nutrition. Functional Plant Biology, 33:153-163.
- Masle J, Gilmore SR, Farquhar GD. 2005 The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature, 436, 866-870
- Buer CS, Wasteneys GO, Masle J. 2003. Ethylene modulates root wave responses in Arabidopsis. Plant Physiology, in press
- Kaiser BN, SR Rawat, Siddiqi MY, Masle J, Glass AD 2002. Functional analysis of an Arabidopsis t-DNA "knock-out" of the high-affinity NH4+ transporter AtAMT1;1. Plant Physiology, 130: 1263-1275.
- Masle J. 2002 Root impedance and plant performance- Physiology, Genetic determinants. In: Plant Roots, The Hidden Half (3rd edition) Y. Waisel, A. Eshel, U. Kafkafi eds, Marcel Dekker, Inc. Publ, NewYork, 807-819.
- Buer S, Masle J, Wasteneys GO. 2001 Growth conditions modulate root-wave phenotypes in Arabidopsis thaliana. Plant and Cell Physiology, 41:1164-1170.
- Masle J. 2000. The effects of elevated [CO2] on cell division rates, growth patterns and blade anatomy in young wheat plants are modulated by factors related to leaf position, vernalisation and genotype. Plant Physiology, 122:1399-1415.
- Masle J. 1999. Root impedance: sensing, signalling and physiological effects. In: Plant responses to environmental stresses: From phytohormones to genome reorganization. H.R. Lerner ed., M. Dekker, Inc., New York Publ., Chapter 22, pp 476-495.
- Masle J. 1998. Growth and stomatal responses of wheat seedlings to spatial heterogeneity of mechanical resistance to root penetration in wheat. Case of bi-layered soils. Journal of Experimental Botany, 49:1245-1257.
- Beemster, GTS, Masle, J, Williamson, RW and Farquhar, GD 1996. Effects of soil resistance to root penetration on leaf expansion. Journal of Experimental Botany, 47, 1663-1678.
- Masle J, Badger MR, Hudson GS. 1993. Effects of ambient CO2 concentration on growth and nitrogen use in tobacco (Nicotiana tabacum) plants transformed with an antisense gene to the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Plant Physiology, 103, 1075-1088.
- Masle, J 1992. Will plant performance on soils prone to drought or with high mechanical impedance to root penetration be improved under elevated atmospheric carbon dioxide? Australian Journal of Botany 40, 491-500.
- Masle, J and Farquhar, GD 1988. Effects of soil strength on the relation of water use efficiency and growth to carbon isotope discrimination in wheat seedlings. Plant Physiology 86, 32-38.